
Fecal Immunochemical Test Interest in Manufacturing and Other Industries
FIT Alternative for Colorectal Cancer Detection
Fecal immunochemical testing (FIT) – a non-invasive screening for colorectal cancer, the second leading cause of cancer death in the United States1 – is garnering increased interest in occupational health care because of two developments. Researchers are learning more about potential environmental and occupational links to the disease, findings which may impact manufacturing and other industries involving widespread use of chemicals.2 The second development is a report by the Centers for Disease Control and Prevention that many Americans are reluctant to have a colonoscopy, the traditional method of colorectal cancer screening. More than a quarter of the general population between the ages of 50 and 75 have never been screened, despite 85 percent being insured.3
Colorectal Cancer: Occupational Associations
Cancer that starts in either the colon (large intestine) or rectum is known as colorectal cancer. It frequently begins with a growth (polyps) on the inner lining in either location. Not all polyps are cancerous; those that are not cancerous are considered benign. Whether a polyp develops into cancer depends on what type it is. Adenomas, or adenomatous polyps, can sometimes change into cancer. Hyperplastic polyps and inflammatory polyps are more common, but in general are not pre-cancerous, according to the American Cancer Society.4
Dietary and lifestyle habits, age, and family history are risk factors traditionally linked to colorectal cancer. It continues to be true that colorectal cancer occurs more frequently in people over age 50, men more than women, and people with a family history of colorectal cancer or certain rare, inherited conditions. Also, people with inflammatory bowel disease and women with ovarian or uterine cancer are more likely to have colorectal cancer.5
In recent years, researchers have begun to see potential associations between colorectal cancer and certain environmental exposures and occupational risk factors. Researchers made an analysis of 83 independent studies (1976 to 2012) to determine if any relationship existed between colorectal cancer and occupations. They concluded industries (such as leather, basic metals, plastic, and rubber manufacturing) with widespread use of chemicals and exposure to asbestos have a higher relative risk of colorectal cancer; however, no consistent pattern was found.2
“Although colorectal cancer, as other tumors or chronic degenerative diseases, is not commonly considered to be occupational (in causation), elevated risks have been reported among workers in some industrial branches such as the textile industry, automobile industry, beverage industry, as well as (employees) exposed to asbestos, dioxin, wood, dust, organic solvents, and metal-working fluids,” investigators noted in summarizing the research studies.
Employees engaged in iron and steel works, dockyards, fur production, tanneries, and petrochemical facilities also were seen as having a higher relative risk for colorectal cancer. Researchers reported a reduced risk of colorectal cancer among employees in crop and agriculture production and some mining activities.
In another study of working-aged men in Japan researchers found a higher risk of colorectal cancer death, among employees in manufacturing and administrative and managerial occupations. In fact, this 2017 study concluded that manufacturing employees and unemployed men, had an 8-to-11-fold greater risk of colorectal, lung and gastric cancer death.6
FIT and Other Colorectal Screening Methods
Generally, there are two types of colorectal cancer screening: visual/structural exams and stool-based tests. Each type includes three different screenings. They are:
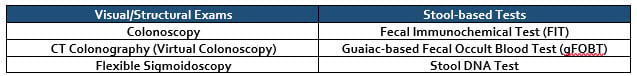
You can read a description of all six screening methods and see a table of general advantages and disadvantages of each on the website of the American Cancer Society.7
Some research points to FIT being superior to the Guaiac-based Fecal Occult Blood Test by having greater participation and fewer false positives.8 Consequently, in this article, for simplicity, we will compare FIT with colonoscopy, the leading visual/structural screening exam.
Colonoscopy is recommended for people who have high-risk factors for colorectal cancer risk, and it also is used in confirmatory testing following positive stool-based tests. People with average risk of colorectal cancer may use either a visual/structural exam or a stool-based test. As a stool-based screen, fecal immunochemical testing looks for microscopic evidence of blood in the stool, a possible early indication of cancer.
Colonoscopy vs. FIT: Key Differences
Invasiveness. A colonoscopy requires insertion of a scope or a tube-like instrument with a light and tiny video camera into a patient’s rectum. This contrasts with a non-invasive FIT screening, which involves a test kit for stool sample collection done in the privacy of the patient’s home.
Advance Preparation. Bowel preparation, including dietary restriction, is needed before a colonoscopy, and use of sedation means the patient needs assistance to get home after the procedure. A FIT screen requires no bowel preparation or dietary restriction ahead of time and no sedation.
Frequency. A standard colonoscopy is recommended once every ten years for adults over age 45 or 50 (depending on medical recommendations). This contrasts with stool-based screening, which is recommended to be done once a year.
Scope. A colonoscopy assesses the entire bowel, compared to a FIT screen’s more limited range of only the lower bowel.
Follow-up to a Positive Result. If clinicians performing a colonoscopy detect polyps, they can remove them at the time of the procedure. When a FIT screen is positive, a colonoscopy is the next step.
Risk. With a colonoscopy, there is a possibility of tears, and bleeding. A FIT screen does not present the same risks.
These differences between the colonoscopy and fecal immunochemical testing may help explain why more than a quarter of the general population, ages 50 to 75, have never been screened for colorectal cancer, despite the vast majority having health insurance.
As researchers learn more about potential occupational links to colorectal cancer, employers may wonder if the FIT screen is as effective as the colonoscopy. Researchers, likewise, have been asking that question.
Fecal Immunochemical Test Effectiveness
Colonoscopy avoidance, due to the procedure’s invasiveness, is creating more interest in FIT as a non-invasive alternative to provide a basic level of colon cancer screening. Is FIT screening worth a closer look for workforce colon cancer protection? Several research studies are generally supportive of fecal immunochemical testing.
A ten-year study published in the New England Journal of Medicine showed that FIT screening and colonoscopy were virtually equivalent in their detection of colorectal cancer; however, the colonoscopy detected adenomas (pre-cancer polyps) more than twice as often as FIT screening. The study also showed that study participants selected for FIT were more willing to get screened. Participants were randomly selected out of a pool of almost 54,000 people to get either a one-time colonoscopy or a FIT screen, once every two years.9
Two other studies, published in 2017 and 2016, also have reported on FIT’s high sensitivity and diagnostic accuracy in detecting colorectal cancer.10,11
The largest clinical trial in Veterans Administration history, called CONFIRM, is soon to deliver results. As another ten-year investigation into the comparative effectiveness of FIT versus colonoscopy for colorectal cancer screening, CONFIRM is due to wrap up in 2020, and offer the latest insight into results for more than 50,000 adults, ages 50 to 75.12,13
Concentra Can Help
If you are interested in learning more about occupational health screenings, including fecal immunochemical testing for colorectal cancer, please contact Concentra through our website.
________________________________________
- Jensen, CD (lead researcher) and 15 others, “Fecal Immunochemical Test Program Performance Over 4 Rounds of Annual Screening: A Retrospective Cohort Study,” Annals of Internal Medicine, April 5, 2016; 164(7): 456-463. Accessed September 14, 2019
- “Occupational exposures and colorectal cancers: A quantitative overview of epidemiological evidence” World Journal of Gastroenterology, September 21, 2014; 20(35): 12431-12444. Accessed September 14, 2019
- “Quick Facts: Colorectal Screening in the United States: Behavioral Risk Factor Surveillance System 2016,” U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Accessed September 14, 2019.
- “What is Colorectal Cancer?” The American Cancer Society. Accessed September 14, 2019
- “Colorectal Cancer: Risk Factors and Prevention,” Cancer.net, November 2018. Accessed September 14, 2019
- Eguchi H, Wada K, Prieto-Merino D, Smith DR, “Lung, gastric, and colorectal cancer mortality by occupation and industry among working-aged men in Japan,” Scientific Reports, February 2017; 7:43204. Accessed September 14, 2019
- “Colorectal Cancer Screening Tests,” American Cancer Society, Accessed on September 14, 2019
- Mousavinezhad M, Majdzadeh R, Akbari Sari A, Delavari A, and Mohtsham F, “The effectiveness of FOBT vs. FIT: A meta-analysis on colorectal cancer screening test,” Medical Journal of the Islamic Republic of Iran, 2016; 30:366. Accessed on September 16, 2019
- Quintero E (lead researcher) and 11 others, “Colonoscopy versus Fecal Immunochemical Testing in Colorectal Cancer Screening,” New England Journal of Medicine, February 23, 2012; 366:697-706. Accessed on September 16, 2019
- Katsoula A, Paschos P, Haidich AB, Tsapas A, and Giouleme O, “Diagnostic Accuracy of Fecal Immunochemical Test in Patients at Increased Risk for Colorectal Cancer: A Meta-analysis, ”JAMA Internal Medicine, August 2017; 177(8):1110-1118. Accessed on September 16, 2019
- Jensen CD (lead researcher) and 15 others, “Fecal Immunochemical Test Program Performance Over Four Rounds of Annual Screening: A Retrospective Cohort Study,” April 2016; 164(7): 456-463. Accessed on September 16, 2019
- Dominitz JA (lead researcher) and 20 others, “Colonoscopy vs. Fecal Immunochemical Test in Reducing Mortality from Colorectal Cancer (CONFIRM): Rationale for Study Design,” American Journal of Gastroenterology, November 2017; 112(11):1736-1746. Accessed September 14, 2019
- U.S. Department of Veterans Affairs, “CONFIRM Now VA’s Largest Clinical Trial Ever.” Accessed on September 14, 2019.



